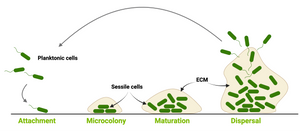
Formation of biofilm negatively influences wound healing and chronic wounds are known to contain biofilms. Advanced anti-biofilm approaches, preventing biofilm formation but also getting-rid-of already formed biofilm are necessary. However, a lack of standardised models for biofilm testing is a barrier in the field. To solve this issue, researchers from University of Tartu, Estonia published a study about novel biofilm models and protocols for assessing the antibacterial and antibiofilm properties of wound dressings. The study by Lorenz et al. showcases an in vitro model prepared by using artificial skin produced by electrospinning and crosslinking a gelatin−glucose matrix and an ex vivo model using pig ear skin. The models were used to study wound dressings with antibacterial properties and evaluated using different types of bacteria (Escherichia coli DSM 1103 (E. coli), Staphylococcus aureus DSM 2569 (S. aureus) and Staphylococcus epidermidis DSM 28319 (S. epidermidis). Extracellular matrix production indicating biofilm formation was shown in skin models of E. coli infections using EbbaBiolight 630. For biofilm quantification, CFU counts were performed after washing planktonic bacteria followed by vortexing and sonification. The work by Lorenz et al. highlights the importance for standardised test systems for biofilm testing as biofilms in wounds are a complex problem that require a holistic picture of the host/material - biofilm - material(wound patch) interface.
Image: Biofilm maturation model on biotic and abiotic surfaces. Created with Biorender. (©Ebba Biotech)
