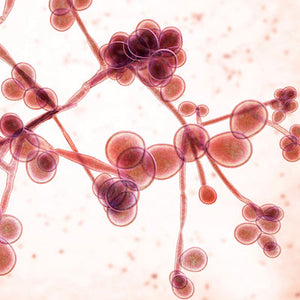
Microbial populations communicate to achieve tasks that a single organism can not. Therefore, understanding the intricate interactions between different microorganisms is an essential missing piece of scientific knowledge. Elucidating the details of the interaction between Pseudomonas aeruginosa and Candida albicans is of particular interest for biochemical science and clinical practice as these pathogens are associated with a high rate of nosocomial infections worldwide. It is known that Pseudomonas and Candida are commonly found together and Pseudomonas is known to reduce the growth of the fungus both via physical contact and the release of chemical signals. However, conventional methods for studying polymicrobial interactions often encounter limitations in precisely discerning specific interactions between microorganisms. In a recent study, researchers from the group of Xiaolong Luo at the Catholic University of America developed a microfluidics platform to try and address this issue. Their innovative approach allowed them to study the complex interplay between Pseudomonas aeruginosa and Candida albicans by growing the bacteria and the fungus in side-by-side microchannels separated by a thin membrane with anti-microbial properties. This approach not only lets the scientists prevent the two species from physically interacting, but also facilitates the continuous monitoring and quantitative analysis of their mutual interactions. Using their system, the researchers were able to confirm that “chemical” contact with Pseudomonas reduces the growth rate of Candida and pushes it towards its hyphal form. However, they also observed how the contact with Candida albicans seems to enhance Pseudomonas growth. At this point, EbbaBiolight 480 was used to confirm that this increased growth is due to the formation of bigger, faster-growing biofilms. Their unique setup and the possibility of using Ebbabiolight 480 in live cultures allowed Pham and colleagues to observe the interaction between Pseudomonas and fully formed Candida albicans hyphae: after 15 hours of co-culture, Pseudomonas busts through the anti-microbial membrane and starts invading the microchannel in which Candida had been growing. In this invasion, the bacteria uses the hyphae as a walkway through the hydrogel matrix, and grows around the fungus, rather than in circular colonies as it normally does. The work from Pham and their colleagues highlights the significance of microbial interactions in invasive infections, as well as the need of better tools to observe these interactions, as the conventional methods could easily be misleading: without the ability to physically separate the two cultures and tracking live the formation of biofilm, observing the way in which Pseudomonas biofilm grows around Candida’s hyphae would not have been possible.
