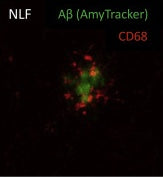
Microglia cells play a vital role in regulating brain development, maintenance of neuronal networks, and injury repair. Their involvement in the progression of protein aggregation leading to neurodegenerative diseases is complex. While they can clear amyloid plaques and prevent their accumulation, dysfunctional microglia regulation may be a key factor in disease progression. Microglia can “sense” accumulation of amyloid plaques which leads to a change in the expression patterns of Plaque Induced Genes (PIGs). One well-known PIG is Trem2, a microglial membrane receptor protein linked with Alzheimer’s disease (AD). A key function of Trem2 is to induce the microglia cells towards a phagocytic phenotype. Therefore, mutations in Trem2 lead to a higher risk of AD. In a study published by Wood et al. in Cell Reports, researchers set out to understand how Trem2 activation works and how it is related to the distance from a plaque. The researchers used spatial transcriptomics to investigate the spatial distribution of gene expression in NLF mice and NLF mice with R47H mutation in Trem2 which is considered a major risk factor for developing AD. To be able to define regions of interest for transcriptomics, Amytracker 520 was used to label plaques. Based on Amytracker 520 signal, radial regions were defined as “on-plaque”, “periplaque” or “away from plaque”. Based on this categorisation, the researchers were able to determine that, from a total of 55 PIGs, 23 (including Trem2) were only upregulated in “on plaque” regions. The significant expression differences between “on-plaque” and “away from plaque” regions observed in mice with functional Trem2 were not apparent in the mice with mutated Trem2. Looking into the functional aspect, the researchers found that, close to plaques, microglia cells express a higher level of CD68 indicative of a phagocytic character. This phenotype was, again, not apparent in mice with mutated Trem2 indicating that the phagocytic phenotypic change of microglia (i.e. CD68 expression) is dependent on the Trem2 genotype. The study highlights the importance of functional microglial activation via Trem2 and the spatial pattern of Trem2 related microglial activation in relation to plaque formation.
Image: Hippocampal section of PFA fixed NLF mouse brain, labelled with Amytracker 520 (green) for amyloid plaques and CD68 (red) indicating the phagocytic phenotype of microglial cells in close proximity to the amyloid plaque. Image from Figure 5C, Wood, J.I. et al. (2022) Cell Reports 41, 111686 (CC BY 4.0).
