The use of Microalgae as a renewable resource has attracted large interest due to their potential for use in the renewable energy, biopharmaceutical, and nutraceutical industries. Microalgae are renewable, sustainable, and economical sources of biofuels, bioactive medicinal products, and food ingredients. Recently, researchers around Professor Avtar Singh Matharu from the Green Chemistry Centre of Excellence in York, United Kingdom have developed a green process to produce cellulose hydrogel using native and spent microalgae. Cellulose extraction from microalgae without any pre-treatment is difficult, since the microalgal cell wall is very thick and rigid, but the researchers have developed a protocol for Microwave-assisted extraction (MAE) which efficiently disrupts algal cell walls aiding the production of defibrillated celluloses that are then further processed to form cellulose-based hydrogels. Cellulose hydrogel is an emerging biomaterial with many different applications such as use as cosmetics, lubricants and within medical biotechnology.
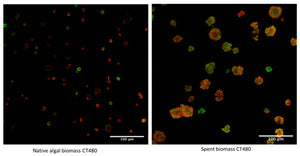
In a recent publication, the researchers at the Green Chemistry Centre of Excellence with Frederick L. Zitzman as lead-author used Carbotrace 480 to benchmark the MAE process and investigate why only MAE at 220°C yields high-quality cellulose hydrogels. Zitzmann et al. performed MAE at different temperatures with native and spent microalgae and used Carbotrace 480 to visualize cellulose in all samples. Carbotrace 480 has its emission maximum in the region of 480 nm. Upon binding to cellulose, the emission peak shifts approx. 20 nm and the intensity in emission increases by around a factor of two. Its spectral properties make Carbotrace 480 the ideal probe for investigating cellulose in microalgae since it allows for spectral unmixing with the autofluorescence of the biomass peaking at 670 nm. Carbotrace 480 shown to be useful to determine differences in morphology between native and spent biomass showing the native biomass as an array of microalgae cells, each with a ring of cellulose encapsulating the cells and the spent biomass as an array of smudged and smashed cells with irregular shapes and a more even distribution of cellulose. Carbotrace 480 also showed clearly an increase in cellulose fluorescence when MAE was performed at higher temperatures in native and spent biomass (see figure). The Carbotrace 480 signal correlated very well with the observed capability for hydrogel formation. Therefore, Carbotrace has proven to be exceptionally suited to aid the exploitation of potential renewable resources and the development of greener processes for hydrogel production.
Image: Carbotrace 480 labeling cellulose in biomass (green) with biomass autofluorescence (red). From Zitzmann, F.L. et al. (2022) Gels, 8(6), 383. (CC BY 4.0)).
